|
9/16/2023 0 Comments Pka to ka 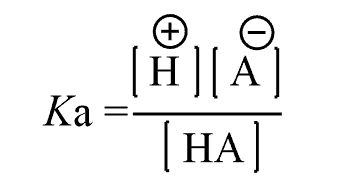 
In this example the pKa on the left is approximately 4 and on the right is 15.7. Therefore, we can compare pKa values of the acid and conjugate acid to determine which side of the equilibrium is favored. Remember, the products of an acid base reaction are the conjugate acid and conjugate base. No doubt about it, going down the hill is much more favorable!!! When Keq < 1 (the less than symbol points to the left, or toward starting materials) energetically the reaction is climbing up the hill to get to products. If Keq 1 (the greater than symbol points to the right towards, or products) energetically the reaction is sliding down the hill to get to products. 
Based on this equation we can conclude that at equilibrium: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
